Your Location:Home >Products >Biochemical Engineering >2018-61-3
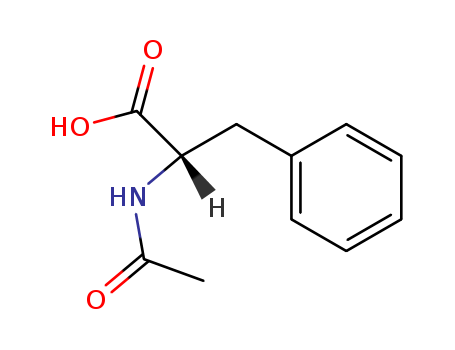

Product Details
|
Chemical Properties |
white to off-white fine cryst. powder or needles |
|
Uses |
It is an essential amino acid produced for medical, feed, and nutritional applications such as in the preparation of aspartame. It is applied as an antidepressant. N-Acetyl-L-phenylalanine can be used as a reactant to synthesize: N-acetyl phenylalanine methyl ester by esterification reaction with methanol using Mukaiyama′s reagent. Acetylaminocyclohexane propanoic acid by rhodium-catalyzed hydrogenation reaction. |
|
Definition |
ChEBI: The N-acetyl derivative of L-phenylalanine. |
|
General Description |
N-Acetyl-L-phenylalanine is an acetyl analog of L-phenylalanine. It is widely used as a reactant to synthesize methyl or ethyl esters of N-acetyl-L-phenylalanine, which are employed as versatile building blocks in peptide synthesis. |
|
Purification Methods |
N-Acetyl-L-phenylalanine is recrystallised from H2O, 20% MeOH/H2O, or CHCl3; dry and store it at 4o. The (DL)-isomer crystallises from H2O, Me2CO, EtOAc, or CHCl3 with m 152-154o and the solubilities in w% at 25o are 0.73 (H2O), 4.3 (Me2CO), 0.79 (EtOAc) and 0.34 (CHCl3) |
InChI:InChI=1/C11H13NO3/c1-8(13)12-10(11(14)15)7-9-5-3-2-4-6-9/h2-6,10H,7H2,1H3,(H,12,13)(H,14,15)/p-1/t10-/m0/s1
Among them, oxidative phosphorylation and phenylalanine metabolism and biosynthesis were triggered by AMF in response to WW and DS, where N-acetyl-L-phenylalanine was …
In this paper, a new multifunctional stationary phase was synthesized by grafting PEI and N-acetyl-L-phenylalanine onto the surface of silica gel, named Sil-PEI-APH.
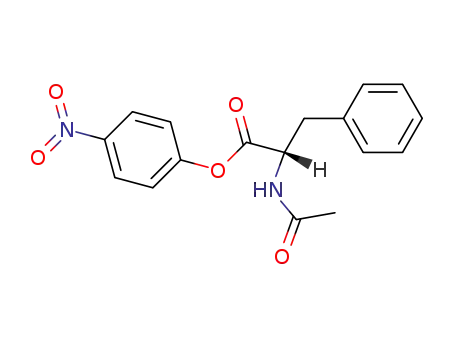
N-acetyl-L-phenylalanine 4-nitrophenyl ester

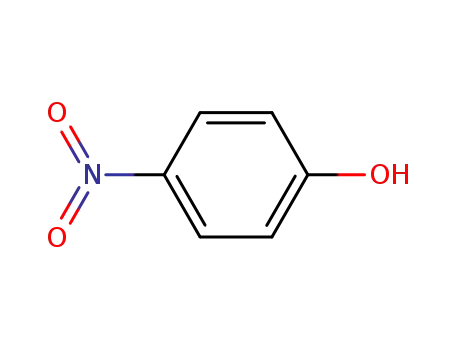
4-nitro-phenol

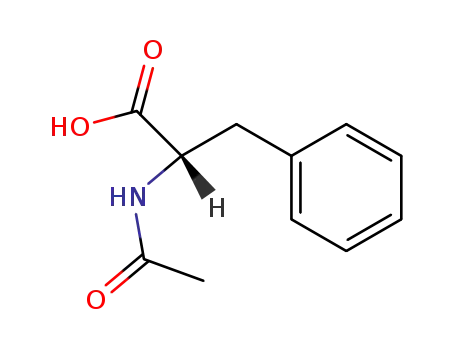
(S)-2-acetylamino-3-phenylpropanoic acid
| Conditions | Yield |
|---|---|
|
PalHis; PalHis + 2C14N2C1Br; In water; acetonitrile; at 25 ℃; Rate constant; furhter bilayer catalytic systems; kcat;
|
|
|
With Nα-tetradecanoyl-L-histidine; In water; acetonitrile; at 10 ℃; Rate constant; Kinetics; Thermodynamic data; ΔH(excit.), ΔS(excit.); Tris-KCl buffer, didodecyldimethylammonium bromide; other nucleophiles and surfactants, other temp.;
|
|
|
With water; poly(iminomethylene); cetylpyridinium chloride; at 23 ℃; Rate constant; pH 5.6; different poly(iminomethylene) 1-cetylpyridinium complexes catalysts and pH-s;
|
|
|
With Z-L-Leu-L-His-L-Leu; Tris buffer; N,N-didodecyl-N,N-dimethylammonium bromide; In acetonitrile; at 24.9 ℃; Rate constant; pH: 7.68, μ = 0.15 (KCl); other peptide catalyst; binding constants Kb/N;
|

1-tetradecyl-3-methylimidazolium L-2-aminohydrocinnamic acid salt

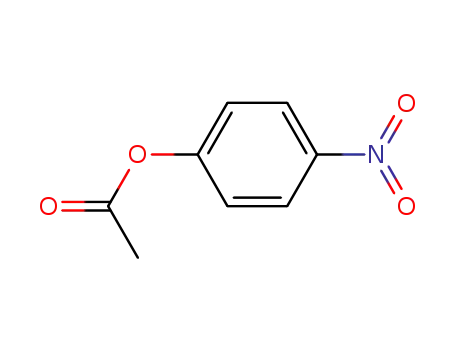
4-nitrophenol acetate


4-nitro-phenol


(S)-2-acetylamino-3-phenylpropanoic acid
| Conditions | Yield |
|---|---|
|
In aq. buffer; at 25 ℃; pH=9; Kinetics;
|
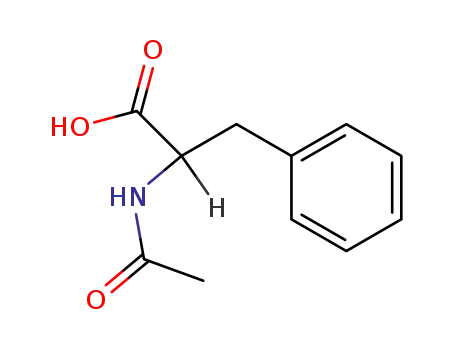
(R,S)-N-acetyl phenylalanine

(1S)-endo-fenchylamine
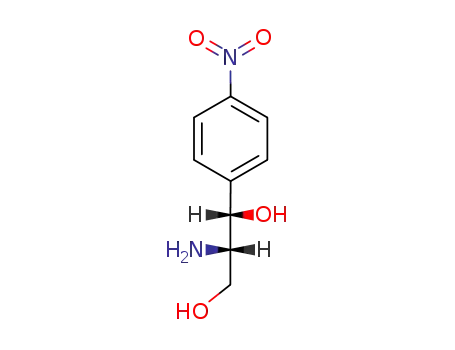
(1S,2S)-2-amino-1-(4-nitrophenyl)propane-1,3-diol
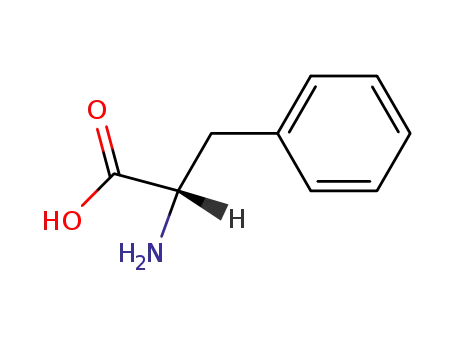
L-phenylalanine

L-phenylalanine
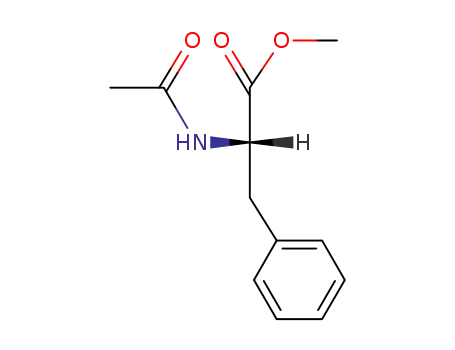
(S)-N-acetylphenylalanine
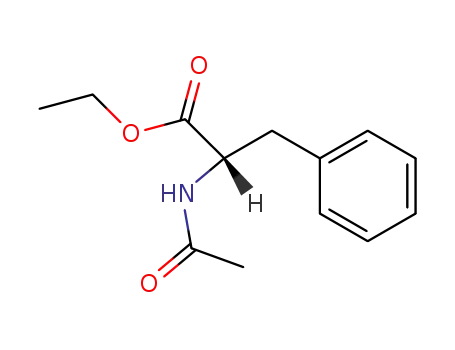
ethyl N-acetyl-(L)-phenylalaninate
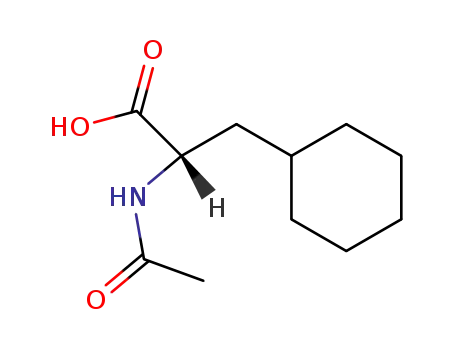
(S)-2-acetylamino-3-cyclohexylpropionic acid
CAS:56-12-2
CAS:7531-52-4
CAS:59-51-8
CAS:1188-21-2